|
|
| Mobile-sized | ||
| This site: | ||
| Home | ||
| Copyright | ||
| Let the Devil Wear Black: | ||
| Briefly. | ||
| ||
| Additional Resources... | ||
| James Linden: | ||
| Background | ||
| Contact | ||
Resources - Extractors
Counter-current solvent extraction in more detail.
Chapter 3
The plant’s effluent had dissolved in it some of the plant’s product as well as many other related compounds and was not really what you wanted dumped into the river so that plants, fish and people downstream could ingest it — this is why dumping the effluent into the river was about to be banned.
However, the harmful part of the effluent — the organic compounds — had a number of properties that could remove it from the effluent so that that could be disposed of in a more responsible manner.
The organic compounds:
- could be broken down into phosphates, chlorides, carbon dioxide and water — the US pilot plant — expensive;
- could be adsorbed onto activated charcoal — drums which had to be monitored constantly and then had to be disposed of — really just transferring the problem to a different medium rather than destroying the compounds; and,
- were extremely soluble in chlorinated organic solvents — such an extraction was already in use on the weekly effluent analysis samples — the resultant mixture with the solvent removed had a market already.
If you took a sample of the effluent and shook it up with a volume of solvent, a certain proportion of the organic compounds would dissolve in the solvent and the rest would remain in the effluent — the concentrations of these gives you the ‘partition’ and the act of mixing the two and letting them separate in equilibrium like that is called a ‘plate’ — analogous to a plate in a distillation column.
To extract a given proportion of the organics from the effluent, you can do a number of things:
- change the ratio of the volumes of effluent and solvent that you use — remember that the partition depends on concentration and not mass;
- change the number of plate–equivalents in the extraction process — you can do this by:
- changing how well it mixes in the extraction process — effectively how many times it gets to mix and then separate before it leaves the process;
- changing the overall flow rates of the solvent and the effluent to change the time that it is in the extractor;
- change the partition — you can only really do this by changing the temperature, the solvent or some other physical property such as acidity of the water–based phase.
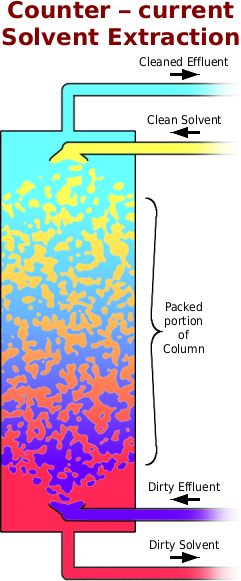
As for the physical reality of the extractor, the diagram pretty much says it all although the packed part of the column is filled with any one of: small hollow cylinders; glass helices; short ceramic tubes; and, so on.
The dirty effluent enters at the distributor at the bottom of the column and floats up onto the underside of the column packing where it breaks up into tiny globules. The clean solvent enters at the distributor at the top of the column and sinks onto the top of the column packing where it breaks up into tiny globules. They then travel through the column, interacting with the other phase until they collect after leaving the packed part of the column and the leave it.
Once it is in equilibrium — both liquids flowing into the column at a constant rate — you get a concentration gradient of the organic compounds as you travel along the extractor.
The compounds are at their highest concentration where the dirty effluent is pumped into the column and as the effluent progresses up the column, they are extracted out of it and end up in the solvent. As the solvent passes down the column, the concentration of the compounds increases as it extracts them from the effluent.
At the top of the packed portion of the column, the cleanest solvent extracts the final amounts of compounds from the cleanest effluent, thus ensuring that it has as much removed from it as possible.
At the bottom, the dirtiest solvent is still able to extract the compounds from the dirtiest effluent because the concentration of compounds in the effluent is still high enough for this to happen.
There is nothing quite like the wrath of a solicitor whose client has been found out.