|
|
| Mobile-sized | ||
| This site: | ||
| Home | ||
| Copyright | ||
| Let the Devil Wear Black: | ||
| Briefly. | ||
| ||
| Additional Resources... | ||
| James Linden: | ||
| Background | ||
| Contact | ||
Resources - EO Unloading
Unloading pressurised tankers without deliberately releasing gasses.
Chapter 9
One of the main issues with the emissions from the plant was the levels of Ethylene Oxide and Propylene Oxide at the perimeter fences being 'ten times the legal limit' as Deryck Simpson put it. The main culprit was venting tanks and reactors.
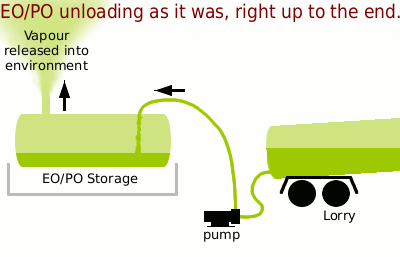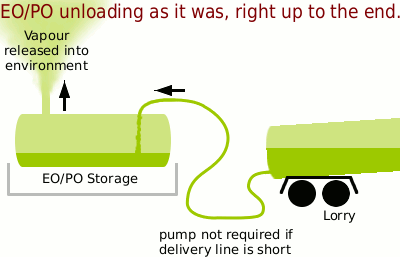
The tanks were kept at 3 atmospheres pressure so that the liquid would not boil by 'padding' the tank with nitrogen — in other words, the tank was pressurised with dry nitrogen to a pressure of 3bar.
The problem was that when a lorry turned up to unload into the tank almost all of the nitrogen padding had to be vented out so as to allow the volume of the incoming liquid into the tank. Additionally, reducing the pressure in the storage tank would allow for speedier unloading.
This caused a great deal of loss of EO or PO to the atmosphere which travelled downwind towards the perimeter fence, tumbling over various buildings and being diluted on the way by the surrounding air but still, being a high enough concentration at the perimeter fence to cause concern — such as getting me to repeat the analysis which only confirmed the original result as being correct.
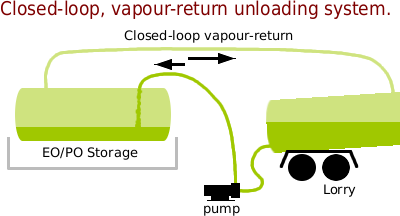
The way to unload EO and PO, keeping releases into the environment to a minimum — realistically, you are still going to end up with some losses as pipes are disconnected and so on — is to take the EO or PO vent gasses and recycle them to the tanker.
The liquid is still pumped out into the storage tanks but there is no continual release of gasses into the environment. This method was known about at the time and was discussed as an alternative at the time so it is not as though it has only been thought of recently.
Was somebody being cautious?